Second Division of Endocrinology, Alexandra Hospital, Athens, Greece
OBJECTIVE: To assess the relationship between circulating 25(OH)D, 1,25(OH)2D, and 24,25(OH)2D in pregnancy at term, and to test the hypothesis that serum 25(OH)D and 24,25(OH)2D are repressed by high serum 1,25(OH)2D levels. DESIGN: Twenty studies in pregnant women at term and 14 studies in young non-pregnant women were selected from the electronic literature. From these studies the mean serum concentrations of 25(OH)D, 1,25(OH)2D, and 24,25(OH)2D from 26 groups of pregnant and 14 groups of non-pregnant young women were analyzed. The grand mean (mean of the means) of serum 25(OH)D, serum 1,25(OH)2D, and of the ratio 1,25(OH)2D/25(OH)D were compared between pregnant and non-pregnant women. Also, the means of serum 1,25(OH)2D were regressed on the means of serum 25(OH)D. The relationship between 25(OH)D and 24,25(OH)2D was also evaluated using similar methods. RESULTS: The grand mean of 1,25(OH)2D was two-fold higher, and the grand mean of the ratio 1,25(OH)2D/25(OH)D was 2.5-fold higher in pregnant than in non-pregnant women with comparable serum 25(OH)D. Regression showed no significant relationship between 1,25(OH)2D and 25(OH)D in pregnant and non-pregnant women. The ratio 24,25(OH)2D/25(OH)D was not different in pregnant and non-pregnanat women. Regression showed significant relationship between 24,25(OH)2D and 25(OH)D in both pregnant and non-pregnant women. CONCLUSIONS: Serum 1,25(OH)2D is not, whereas serum 24,25(OH)2D is significantly related to serum 25(OH)D in pregnant women. Serum 24,25(OH)2D seems not to be repressed by the high 1,25(OH)2D levels during pregnancy, whereas a similar conclusion for 25(OH)D is less clear.
Pregnancy, Vitamin D deficiency, Vitamin D metabolites
INTRODUCTION
Humans usually obtain more than 80% of their vitamin D3 from their skin where it is synthesized through the action of ultraviolet sunlight, acquiring the rest from foodstuffs (mainly oily fish or vitamin D fortified food products). The molecule of vitamin D (D3 or D2) is hydroxylated in the liver and converted to 25-hydroxyvitamin D [25(OH)D], which in turn undergoes a second hydroxylation in the kidney (and many other tissues) resulting in the formation of 1,25-dihydroxyvitamin D [1,25(OH)2D], the biologically active metabolite of vitamin D.
The regulation of the production and catabolism of 1,25(OH)2D has recently been reviewed by Plum and DeLuca.1 Most, if not all, of the circulating 1,25(OH)2D is produced in the kidney through the action of the enzyme 1-α-hydroxylase (CYP27B1) on 25(OH)D. Hypocalcemia activates the secretion of PTH, which acting on the kidney induces the activity of renal 1-α-hydroxylase leading to increased circulating levels of 1,25(OH)2D. Hypophosphatemia can also activate renal 1-α-hydroxylase by an non-clear mechanism, leading also to increased circulating 1,25(OH)2D. When both serum calcium and serum phosphorus concentrations are low, renal 1-α-hydroxylase is super-stimulated, resulting in extremely high circulating 1,25(OH)2D.1 The catabolism of 1,25(OH)2D is also regulated to prevent hypercalcemia and hyperphosphatemia.1 The main catabolic enzyme in the vitamin D endocrine system is 24-hydroxylase (CYP24A1). This enzyme attenuates the vitamin responsiveness by catalyzing synthesis of the inactive metabolites 1,24,25 (OH)3D and 24,25(OH)2D from 1,25(OH)2D and 25(OH)D, respectively. The high PTH during hypocalcemia not only stimulates the production of 1,25(OH)2D but also decreases its catabolism by decreasing 24-hydroxylase activity, thereby repressing the production of 1,24,25 (OH)3D and 24,25(OH)2D. Low serum phosphorus decreases the mRNA levels of the 24-hydroxylase to prevent catabolism of 1,25(OH)2D. Transcription of the 24-hydroxylase gene is stimulated by 1,25(OH)2D and thus 1,25(OH)2D enhances its own catabolism, while PTH has a key control of 24-hydroxylase activity.1
The best marker of vitamin D status in the adult is considered to be the 25(OH)D level. Serum concentrations of 25(OH)D less than 50 nmol/L characterize vitamin D insufficiency, and less than 20 nmol/L severe vitamin D deficiency. The critical 25(OH)D value of 50 nmol/L is based on the finding that after treatment of vitamin D deficient persons with stepwise increased doses of vitamin D, no further suppression of plasma parathyroid hormone (PTH) was noted when serum level of 25(OH)D surpassed 50 nmol/L.2 From another point of view, on using regression of plasma PTH on serum 25(OH)D, it was observed that plasma PTH started to rise when serum 25(OH)D became lower than 75 nmol/L; thus, serum 25(OH)D values below this critical level may cause secondary hyperparathyroidism and have been considered to be diagnostic of vitamin D insufficiency.3,4 The 25(OH)D cut-off value of 80 nmol/L is corroborated by the work of Heany5 who found that 25(OH)D values of 80 nmol/L or higher are needed for optimal intestinal absorption of calcium. Further validation of the 75 nmol/L serum concentration as a lower limit of vitamin D sufficiency originated from clinical trials for the treatment of vitamin D deficiency. In several recent trials it was demonstrated that treatment of vitamin D deficient elderly subjects with vitamin D supplements significantly decreased the risk for hip fracture only when 25(OH)D levels of at least 74 nmol/L were achieved during therapy.6 Finally, Rejmark et al7 demonstrated that in postmenopausal women serum 25(OH)D is the major determinant of serum levels of 1,25(OH)2D; at 25(OH)D serum levels above 80 nmol/L, serum 1,25(OH)2D do not increase further, whereas below this 25(OH)D level, serum 1,25(OH)2D decreases progressively with decreasing 25(OH)D levels.
Several longitudinal8-11 and cross-sectional12-14 studies have shown that, when compared to non-pregnant young women, pregnant women have high serum 1,25(OH)2D concentration from early pregnancy; serum 1,25(OH)2D rises steadily throughout gestation and reaches levels about double those of non-pregnant women at term. The contribution of the placenta to the high circulating 1,25(OH)2D is not clear. Human placenta and decidua can synthesize 1,25(OH)2D. Vitamin D receptors (VDR), 1-α-hydroxylase, and 24-hydroxylase are expressed in human placenta and decidua.15 Zehnder et al15 demonstrated that the expression of mRNA for 1-α-hydroxylase in decidua was ~1000-fold higher in the first and second trimester when compared with the third trimester of human pregnancy. In placenta, 1-α-hydroxylase expression was 80-fold higher in the first and second when compared with the third trimester biopsies. Therefore, if the placenta were the main source of circulating 1,25(OH)2D one would expect the level of 1,25(OH)2D to be higher during early than late stages of pregnancy, unless the catabolism of 1,25(OH)2D during early pregnancy is much higher than in advanced gestation. Thus, it seems more probable that the 1,25(OH)2D in the maternal circulation originates mainly from the maternal kidneys (see also review by Kovacs).16 Plasma concentration of vitamin D binding protein (DBP) is high in pregnancy13 and this, at least partly, accounts for the frankly elevated total 1,25(OH)2D; free serum 1,25(OH)2D was found to be high13,17 or normal18 during pregnancy (see ref. 18 for discussion on free serum 1,25(OH)2D in pregnancy).
The high 1,25(OH)2D serum concentration could theoretically induce some changes in the metabolism of vitamin D during human pregnancy. Halloran and Castro19 found that chronic 1,25(OH)2D administration in the rat reduced the serum concentration of 25(OH)D by increasing its metabolic clearance rate (MCR) without affecting its production rate (PR). This treatment also increased both the MCR and PR of 24,25(OH)2D. However, the disproportionately larger increase in MCR resulted also in a decrease in the serum concentration of 24,25(OH)2D.19 The findings of Berlin and Björkhem were similar.20 Clements et al21 showed that in rats the hepatic catabolism of 25(OH)D is increased by calcium deprivation and that this effect is mediated by 1,25(OH)2D produced in response to secondary hyperparathyroidism. Administration of 1,25(OH)2D in humans significantly decreased serum 25(OH)D concentration.22 Davies et al23 found a significant negative relationship between the elimination half-time (t½) of [3H]25(OH)D3 in serum and serum 1,25(OH)2D concentration in patients with partial gastrectomy and elevated 1,25(OH)2D, a finding implying that the endogenous high serum 1,25(OH)2D increased the catabolism of 25(OH)D in these patients. Clements et al24 showed that the t½ of [3H]25(OH)D3 in serum was shortened by raising the serum concentration of 1,25(OH)2D in patients with various disorders of bone and mineral metabolism.
The aim of the present meta-analysis was to evaluate the relationship between serum 25(OH)D and serum 1,25(OH)2D as well as between serum 25(OH)D and serum 24,25(OH)2D in pregnant women, and also to test the hypothesis that in human pregnancy the high 1,25(OH)2D serum concentration may be causing increased catabolism of 25(OH)D and thus resulting in relatively low levels of serum 25(OH)D.
METHODS
The electronic literature (Medline) was searched using the MeSH terms “pregnancy” and “vitamin D” for studies in pregnant women in which measurements of vitamin D metabolites were included. Twenty studies, comprising 26 groups of pregnant women at term, in which at least two of the vitamin D metabolites 25(OH)D, 1,25(OH)2D, and 24,25(OH)2D were determined, were selected for analysis. The pregnant women of 19 groups did not take vitamin supplements (Table 1A ). The women of 7 groups were supplemented with vitamin D (400 IU/d) during their pregnancy (Table 1B ). Fourteen control groups of young healthy non-pregnant women, of which 9 were part of the pregnancy studies and 5 were from independent studies, were also analyzed (Table 1C ). The means of the parameters from each group of women were analyzed. In the 20 groups of pregnant and 9 groups of non-pregnant women measurements of both 1,25(OH)2D and 25(OH)D were available and thus the relationship of these two metabolites could be evaluated. In the 13 groups of pregnant and 7 groups of non-pregnant women results of both 25(OH)D and 24,25(OH)2D were available for a similar analysis.
Comparison between two means by t test and linear regression of the means of a vitamin D metabolite on the means of a second vitamin D metabolite were performed using GraphPad Prism, GraphPad Software, San Diego, California, USA. Significance was set as a two-tailed p<0.05.
RESULTS
The mean of the means (grand mean) of serum 25(OH)D, serum 1,25(OH)2D, and serum 24,25(OH)2D were not significantly different between vitamin D supplemented and unsupplemented pregnant women and thus the two categories of pregnant women were combined into a single category.
The relationship between 1,25(OH)2D and 25(OH)D is shown in Figure 1. The mean of the means (grand mean) of serum 25(OH)D in pregnant women (n=20 means) was not different from the grand mean (n= 9 means) in non-pregnant controls (48.2±5.6 nmol/L vs 51.8±6.1, p=0.71). In the same groups, the grand mean of serum 1,25(OH)2D was significantly higher in pregnant women than in the controls (186.7± 13.5 pmol/L vs 91.4±4.2, p<0.0001). The ratio 1,25(OH)2D/25(OH)D was calculated by dividing the mean 1,25(OH)2D serum value (pmol/L) by the mean 25(OH)D serum value (nmol/L) in each group. The grand mean of the 1,25(OH)2D/25(OH)D ratio was significantly higher in the pregnant women than in the controls (4.9±0.64 vs 1.96±0.23, p=0.006). In order to determine whether high serum 1,25(OH)2D represses serum 25(OH)D during pregnancy, the serum 1,25(OH)2D means were regressed on the serum 25(OH)D means. There was no significant relationship between 1,25(OH)2D and 25(OH)D in pregnant women (R2=0.08, p=0.22) or in non-pregnant controls (R2=0.001, p=0.93) (Figure 1). In the majority of the groups of pregnant women at term the means of serum 1,25(OH)2D were found to be between 138 and 242 pmol/L, with the corresponding means of serum 25(OH)D between 15 and 82 nmol/L. In the groups of non-pregnant women the means of serum 1,25(OH)2D were between 71 and 108 pmol/L, with the corresponding means of serum 25(OH)D between 25 and 88 nmol/L (Figure 1).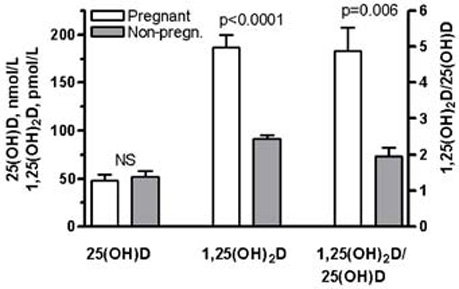

Figure 1. Comparison of the grand means (±SEM) of serum 25(OH)D (nmol/L), serum 1,25(OH)2D (pmol/L), and of the ratio 1,25(OH)2D/25(OH)D between pregnant (n=20) and non-pregnant (n=9) young women (Α). Linear regression of the serum 1,25(OH)2D means on the serum 25(OH)D means in pregnant (n=20) and non-pregnant women (n=9) (Β). NS=Non significant.
The relationship between 24,25(OH)2D and 25(OH)D is shown in Figure 2. The serum 25(OH)D grand mean in the pregnant women (n=13 means) was significantly lower than the serum 25(OH)D grand mean (n=7 means) in the controls (48.5± 4.1 nmol/L vs 64.9±6.3, p=0.036). In the same groups, the grand means of 24,25(OH)2D and of the ratio 24,25(OH)2D/25(OH)D were not different between pregnant and non-pregnant women (3.9±0.4 nmol/L vs 5.0±0.7, p=0.60) and (0.081±vs 0.076, p=0.12), respectively. Linear regression of the serum 24,25(OH)2D means on the serum 25(OH)D means showed significant positive relationship between the two metabolites in the pregnant women (R2=0.42, p=0.016), and almost significant relationship between the two metabolites in the controls (R2=0.49, p=0.08). The slope of the two regression lines was identical and, in fact, the two lines were virtually superimposed (Figure2).
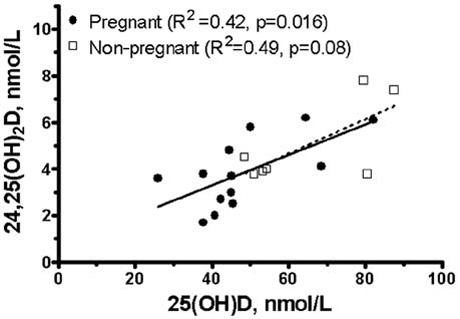
Figure 2. Comparison of the grand means (±SEM) of serum 25(OH)D, serum 24,25(OH)2D, and of the ratio 24,25(OH)2D/25(OH)D between pregnant (n=13) and non-pregnant (n=7) young women (A). Linear regression of the serum 24,25(OH)2D means on the serum 25(OH)D means in pregnant (n=13) and non-pregnant women (n=7) (B). NS=Non significant.
DISCUSSION
High 1,25(OH)2D serum levels after administration of 1,25(OH)2D in rats19,20 or in humans22 have been shown to cause a reduction in the serum level of 25(OH)D by increasing its metabolic clearance rate19 or reducing its t½.24 Increased catabolism of 25(OH)D was demonstrated after inducing high levels of endogenous 1,25(OH)2D by low calcium diet in rats.21 Clements et al24 studied patients with various disorders of bone and mineral metabolism and demonstrated that raising serum 1,25(OH)2D reduces the elimination half-time of 25(OH)D in serum, which means that high 1,25(OH)2D accelerates the catabolism of 25(OH)D. The findings of Davies 23 were similar in patients with partial gastrectomy and secondary hyperparathyroidism with high 1,25(OH)2D serum levels.
One of the objectives of this meta-analysis was to investigate if the same effect of high 1,25(OH)2D serum levels upon serum 25(OH)D occurs in human pregnancy. In the 20 groups of pregnant and 9 groups of non-pregnant women, both mean serum 25(OH)D and mean serum 1,25(OH)2D were available for analysis. While the grand mean (mean of the means of each group) of serum 25(OH)D in pregnant women was not statistically different from the grand mean in non-pregnant women, the grand mean of serum 1,25(OH)2D in pregnant women was twice as high when compared to the grand mean of non-pregmant women. The grand mean of the 1,25(OH)2D/25(OH)D ratio was approximately 2.5 times higher in pregnant women when compared to non-pregnant women. These data, taken together, imply that in pregnant women at term a frankly high level of serum 1,25(OH)2D is maintained in comparison to non-pregnant women despite non-different 25(OH)D serum levels (Figure 1). It is not possible from the data analyzed here to determine if the high serum level of 1,25(OH)2D is exclusively due to increased binding of the hormone to DBP or also to a concomitant increase of free 1,25(OH)2D. Regression of the means of serum 1,25(OH)2D on the means of serum 25(OH)D showed no significant relationship between the two metabolites in pregnant women. This finding militates against a possible repressing effect of high serum 1,25(OH)2D on serum 25(OH)D in pregnancy. Also, in favor of this opinion is the fact that serum 25(OH)D does not seem to rise after delivery when serum 1,25(OH)2D falls abruptly.9,10 No significant relationship was also found between the means of serum 1,25(OH)2D and the means of serum 25(OH)D in the young non-pregnant women. This finding differs from that of Rejnmark et al7 in postmenopausal women and raises the question whether the relationship between serum 1,25(OH)2D and serum 25(OH)D may be age dependent.
High serum 1,25(OH)2D also induced a fall in serum 24,25(OH)2D in rats, in parallel with the fall in serum 25(OH)D.19 In patients with partial gastrectomy and high serum 1,25(OH)2D, serum 24,25(OH)2D was found to be significantly lower than in controls.44 In the 13 groups of pregnant and 7 groups of non-pregnant women, both mean serum 25(OH)D and mean serum 24,25(OH)2D were available for analysis. The grand mean of serum 25(OH)D was significantly lower in pregnant than in control women. The grand mean of serum 24,25(OH)2D was also lower in the pregnant women, although not significantly. However, the grand means of the 24,25(OH)2D/25(OH)D ratio in pregnant and control women were not different (Figure 2). Regression of the means of serum 24,25(OH)2D on the means of serum 25(OH)D showed a significant relationship between the two vitamin D metabolites in pregnant women and almost significant relationship in the controls. The regression coefficients indicated that 42% of the variation of the means of serum 24,25(OH)2D could be explained by changes in the means of serum 25(OH)D in pregnant women, and 49% of the variation of the means of serum 24,25(OH)2D could be explained by changes in the means of serum 25(OH)D in non-pregnant women (Figure 2). These data imply that very likely the metabolism of 24,25(OH)2D is not different in pregnant and non-pregnant women.
The substrate for the biosynthesis of both 1,25(OH)2D and 24,25(OH)2D is 25(OH)D.1 This meta-analysis showed that serum 1,25(OH)2D is not related to serum 25(OH)D, whereas by contrast serum 24,25(OH)2D is significantly related to serum 25(OH)D in both pregnant and non-pregnant young women. These findings allow the conclusion that in human pregnancy the high serum levels of 1,25(OH)2D do not seem to induce changes in the metabolism of 24,25(OH)2D, while for a possible effect of high 1,25(OH)2D on 25(OH)D a similar conclusion is less clear. The best method to resolve this matter is probably the measurement of the elimination half-time of serum 25(OH)D in pregnant women, using the method employed by Clements et al20,24
In the majority of the studies analyzed here, mean serum 1,25(OH)2D in pregnant women was high (>138 pmol/L), while mean serum 25(OH)D was less than 50 nmol/L, and in three studies less than 25 nmol/L. Only in one study30 did pregnant women with severe vitamin D deficiency [extremely low 25(OH)D] have relatively low serum 1,25(OH)2D. Consequently, it seems uncertain whether many pregnant women with low serum 25(OH)D but high serum 1,25(OH)2D may be characterized as vitamin D deficient. On the other hand, among the studies herein analyzed, only in one study34 was mean serum 25(OH)D above 80 nmol/L and in this case mean serum 1,25(OH)2D was not unusually high for pregnant women (see Table 1 and Figure 1). Therefore, it is not possible from these data to predict 1,25(OH)2D serum levels when 25(OH)D serum levels are frankly above 80 nmol/L during pregnancy.
Among the limitations of this meta-analysis are the limited number of the analyzed studies, the heterogeneity of the studied populations, and the variable methods used for the measurements of the vitamin D metabolites.
Possible mechanisms responsible for the high serum 1,25(OH)2D in pregnancy have been proposed (see review by Kovacs).16 Theoretically, super-stimulation of renal 1-α-hydroxylase1 by an undefined mechanism can result in extremely high circulating 1,25(OH)2D in the pregnant woman. Another possibility, not excluding the previous one, is increased availability of 25(OH)D to the maternal renal 1-α-hydroxylase for activation. How can this be accomplished even in cases with relatively low levels of 25(OH)D in the maternal circulation? A major pathway by which 25(OH)D can enter the epithelial cells of the proximal renal tubules for activation is through their luminal site. Nykjaer et al45 have demonstrated that the filtered DBP-25(OH)D complex is taken up from the glomerular filtrate by the epithelial cells of the proximal renal tubules through endocytic megalin-cubilin coreceptors. Glomerular filtration rate is increased in pregnant women and this may theoretically result in increased filtered load of DBP-25(OH)D and increased uptake of the complex by the renal tubular epithelium. The existence of this pathway implies that the free hormone hypothesis is not essential to explain the uptake of 25(OH)D or 1,25(OH)2D by cells of various tissues having megalin receptors.45
In two longitudinal studies,10,46 the true percentage of calcium absorption was found to be higher in pregnant women with low46 compared to pregnant women with high calcium intake.10 Then again, urinary excretion of calcium increased during pregnancy in women with high calcium intake,10 but decreased in women with low calcium intake.46 Therefore, it seems that calcium homeostasis can adapt to low calcium intake during pregnancy.46 The mechanism for this adaptation is not understood, and the role of the high 1,25(OH)2D serum levels in the increased intestinal absorption of calcium during pregnancy has not been determined.16,46 Many hormones such as PTH, estrogens, prolactin, and human placental lactogen may apparently be involved in the regulation of calcium homeostasis in pregnancy, while it has been suggested that vitamin D does not have an important role in the increased intestinal calcium absorption during pregnancy.16 The possibility that the hypercortisolism of pregnancy47 may be a cause of some degree of vitamin D resistance and the effects on calcium homeostasis of the chronic respiratory alkalosis48 commonly affecting pregnant women should also be considered.
In conclusion, it is not clear whether vitamin D deficiency [low 25(OH)D serum levels] is more frequent in pregnant women when compared to age matched non-pregnant women. This may be very difficult to determine since vitamin D status depends on a multitude of environmental factors. It could be argued that pregnant women may be characterized as having hypervitaminosis D because of their frankly elevated 1,25(OH)2D serum concentration. This meta-analysis demonstrated that the elevated serum 1,25(OH)2D during pregnancy is not related to serum 25(OH)D. It also showed that, very likely, the high serum 1,25(OH)2D in pregnancy does not repress the serum 25(OH)D levels as was demonstrated in studies in animals and non-pregnant humans. Paraphrasing Kovacs,16 we consider that there is need for prospective studies with large numbers of pregnant women to elucidate whether the same criteria established for the definition of vitamin status in the non-pregnant adults can also be applied to pregnant women.
REFERENCES
1. Plum LA, DeLuca HF, 2009 The functional metabolism and molecular biology of vitamin D action. Clinic Rev Bone Miner Metab 7: 20-21.
2. Malabanan A, Veronikis IE, Holick MF, 1998 Redefining vitamin D insufficiency. Lancet 351: 805-806.
3. Chapuy MC, Preziosi P, Maamer M, et al, 1997 Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos Int 7: 439-444.
4. Papapetrou PD, Triantaphyllopoulou M, Karga H, et al, 2007 Vitamin D deficiency in the elderly in Athens, Greece. J Bone Miner Metab 25: 198-203.
5. Heaney RP, 2004 Functional indices of vitamin D status and ramifications of vitamin D deficiency. Am J Clin Nutr 80: Suppl 6:1706-1709.
6. Bischoff-Ferrari HA, Willett WC, Wong JB, Giovannucci E, Dietrich T, Dawson-Hughes B, 2005 Fracture prevention with vitamin D supplementation: a meta-analysis of randomized controlled trials. JAMA 293: 2257-2264.
7. Rejnmark L, Vestergaard P, Heickendorff L, Mosekilde L, 2008 Plasma 1,25(OH)2D levels decrease in postmenopausal women with hypovitaminosis D. Eur J Endocrinol 158: 571-576.
8. Seki K, Makimura N, Mitsui C, Jirata J, Nagata I, 1991 Calcium-regulating hormones and osteocalcin levels during pregnancy: a longitudinal study. Am J Obstetr Gynecol 164: 1248-1252.
9. Ardawi MSM, Nasrat HAN, BA’Aqueel HS, 1997 Calcium-regulating hormones and parathyroid hormone-related peptide in normal human pregnancy and postpartum: a longitudinal study. Eur J Endocrinol 137: 402-409.
10. Ritchie LD, Fung EB, Halloran BP, et al, 1998 A longitudinal study of calcium homeostasis during human pregnancy and lactation and after resumption of menses. Am J Clin Nutr 67: 693-701.
11. Uemura H,Yasui T, Kiyokawa M, et al, 2002 Serum osteoprotegerin/osteogenesis-inhibitory factor during pregnancy and lactation and the relationship with calcium-regulating hormones and bone turnover markers. J Endocrinol 174: 353-359.
12. Whitehead M, Lane G, Young O, et al, 1981 Interrelations of calcium-regulating hormones during normal pregnancy. Br Med J (Clin Res Ed) 283: 10-12.
13. Bouillon R, Van Assche FA, Van Baelen H, Heyns W, De Moor P, 1981 Influence of the vitamin D-binding protein on the serum concentration of 1,25-dihydroxyvitamin D3. Significance of the free 1,25-dihydroxyvitamin D3 concentration. J Clin Invest 67: 589-596.
14. Kuoppala T, Tuimala R, Parviainen M, Koskinen T, Ala-Houhala M, 1986 Serum levels of vitamin D metabolites,calcium, phosphorus, magnesium and alkaline phosphatase in Finnish women throughout pregnancy and in cord serum at delivery. Hum Nutr Clin Nutr 40: 287-293.
15. Zehnder D, Evans KN, Kilby MD, et al, 2002 The ontogeny of 25-hydroxyvitamin D3 1α-hydroxylase expression in human placenta and decidua. Am J Pathol 161: 105-114.
16. Kovacs CS, 2008 Vitamin D in pregnancy and lactation:maternal, fetal, and neonatal outcomes from human and animal studies. Am J Clin Nutr 88: Suppl: 520-528.
17. Bikle DD, Gee E, Halloran B, Haddad JG, 1984 Free 1,25-dihydroxyvitamin D levels in serum from normal subjects, pregnant subjects, and subjects with liver disease. J Clin Invest 74: 1966-1971.
18. van Hoof HJC, de Sévaux RGL, van Baelen H, et al, 2001 Relationship between free total 1,25-dihydroxyvitamin D in conditions of modified binding. Eur J Endocrinol 144: 391-396.
19. Halloran BP, Castro ME, 1989 Vitamin D kinetics in vivo: effect of 1,25-dihydroxyvitamin D administration. Am J Physiol 256: E686-E691.
20. Berlin T, Björkhem I, 1987 On the regulatory importance of 1,25-dihydroxyvitamin D3 and dietary calcium on serum levels of 25-hydroxyvitamin D3 in rats. Biochem Biophys Res Commun 144: 1055-1058.
21. Clements MR, Johnson L, Fraser DR, 1987 A new mechanism for induced vitamin D deficiency in calcium deprivation. Nature 325: 62-65.
22. Loré F, Di Cairano G, Periti P, Caniggia A, 1982 Effect of the administration of 1,25-dihydroxyvitamin D3 on serum levels of 25-hydroxyvitamin D in postmenopausal women. Calcif Tissue Int 34: 539-541.
23. Davies M, Heys SE, Selby PL, Berry JL, Mawer EB, 1997 Increased catabolism of 25-hydroxyvitamin D in patients with partial gastrectomy and elevated 1,25-dihydroxyvitamin D levels. Implications for metabolic bone disease. J Clin Endocrinol Metab 82: 209-212.
24. Clements MR, Davies M, Hayes ME, et al, 1992 The role of 1,25-dihydroxyvitamin D in the mechanism of acquired vitamin D deficiency. Clin Endocrinol 37: 17-27.
25. Fleischman AR, Rosen JF, Cole J, Smith CM, DeLuca HF, 1980 Maternal and fetal serum 1,25-dihydroxyvitamin D levels at term. J Pediatr 97: 640-642.
26. Gertner JM, Glassman MS, Coustan DR, 1980 Goodman BP. Fetomaternal vitamin D relationships at term. J Pediatr 97: 637-640.
27. Delvin EE, Glorieux FH, Salle BL, David L, Varenne JP, 1982 Control of vitamin D metabolism in preterm infants: feto-maternal relationships. Arch Dis Child 57: 754-757.
28. Seino Y, Ishida M, Yamaoka K, et al, 1982 Serum calcium regulating hormones in the perinatal period. Calcif Tissue Int 34: 131-135.
29. Shany S, Biale Y, Zuili I, Yankowitz N, Berry JL, Mawer EB, 1984 Feto-maternal relationships between vitamin D metabolites in Israeli Bedouins and Jews. Am J Clin Nutr 40: 1290-1294.
30. Mallet E, Gugi B, Brunelle P, Henocq A, Basuyau JP, Lemeur H, 1986 Vitamin D supplementation in pregnancy: a controlled trial of two methods. Obstet Gynecol 68: 300-304.
31. Ron M, Menczel J, Schwartz L, Palti Z, Kidroni G, 1987 Vitamin D3 metabolites in amniotic fluid in relation with maternal and fetal sera in term pregnancies. J Perinat Med 15: 282-290.
32. Nehama H, Wientroub S, Eisenberg Z, Birger A, Milbauer B, Weisman Y, 1987 Seasonal variation in paired maternal-newborn serum 25-hydroxyvitamin D and 24,25-dihydroxyvitamin D concentrations in Israel. Isr J Med Sci 23: 274-277.
33. Brunvand L, Haug E, 1993 Vitamin D deficiency amongst Pakistani women in Oslo. Acta Obstet Gynecol Scand 72: 264-268.
34. Seely EW, Brown EM, DeMaggio DM, Weldon DK, Graves SW, 1997 A prospective study of calciotropic hormones in pregnancy and post partum: reciprocal changes in serum intact parathyroid hormone and 1,25-dihydroxyvitamin D. Am J Obstet Gynecol 176: 214-217.
35. Weisman Y, Occhipinti M, Knox G, Reiter E, Root A, 1978 Concentrations of 24,25-dihydroxyvitamin D and 25-hydroxyvitamin D in paired maternal-cord sera. Amer J Obstet Gynecol 130: 704-707.
36. Reiter EO, Braunstein GD, Vargas A, Root AW, 1979 Changes in 25-hydroxyvitamin D and 24,25-dihydroxyvitamin D during pregnancy. Am J Obstet Gynecol 135: 227-229.
37. Markestad T, Aksnes L, Ulstein M, Aarskog D, 1984 25-Hydroxyvitamin D and 1,25-dihydroxyvitamin D of D2 and D3 origin in maternal and umbilical cord serum after vitamin D2 supplementation in human pregnancy. Am J Clin Nutr 40: 1057-1063.
38. Hollis BW, Pittard WB 3rd, 1984 Evaluation of the total fetomaternal vitamin D relationships at term: evidence for racial differences. J Clin Endocrinol Metab 59: 652-657.
39. Kano K, Yoshida H, Yata J, Suda T, 1980 Age and seasonal variations in the serum levels of 25-hydroxyvitamin D and 24,25-dihydroxyvitamin D in normal humans. Endocrinol Jpn 27: 215-221.
40. Parviainen MT, Savolainen KE, Alhava EM, Mäenpää PH, 1981 5-hydroxyvitamin D2, 25-hydroxyvitamin D3 and total 24,25-dihydroxyvitamin D in human serum. Ann Clin Res 13: 26-33.
41. Van Der Klis FR, Jonxis JH, Van Doormaal JJ, Sikkens P, Saleh AE, Muskiet FA, 1996 Changes in vitamin-D metabolites and parathyroid hormone in plasma following cholecalciferol administration to pre- and postmenopausal women in the Netherlands in early spring and to postmenopausal women in Curaηao. Br J Nutr 75: 637-646.
42. Kinyamu HK, Gallagher JC, Balhorn KE, Petranick KM, Rafferty KA, 1997 Serum vitamin D metabolites and calcium absorption in normal young and elderly free-living women and in women living in nursing homes Am J Clin Nutr 65:790-797. Erratum in: Am J Clin Nutr 66: 454
43. Levis S, Gomez A, Jimenez C, et al, 2005 Vitamin D deficiency and seasonal variation in an adult South Florida population. J Clin Endocrinol Metab 90: 1557-1562.
44. Klein KB, Orwoll ES, Lieberman DA, Meier DE, McClung MR, Parfitt AM, 1987 Metabolic bone disease in asymptomatic men after partial gastrectomy with Billroth II anastomosis. Gastroenterology 92: 608-616.
45. Nykjaer A, Fyfe JC, Kozyraki R, et al, 2001 Cubilin dysfunction causes abnormal metabolism of the steroid hormone 25(OH) vitamin D3. Proc Natl Acad Sci USA 98: 13895-13900.
46. Zapata CLV, Domangelo CM, Woodhouse LR, Abrams SA, Spencer EM, King JC, 2004 Calcium homeostasis during pregnancy and lactation in Brazilian women with low calcium intakes: a longitudinal study. Am J Clin Nutr 80: 417-422.
47. Clerico A, Del Chicca MG, Ferdeghini M, Ghione S, Materazzi F, 1980 Progressively elevated levels of biologically active (free) cortisol during pregnancy by a direct radioimmunological assay of diffusible cortisol in an equilibrium dialysis system. J Endocrinol Invest 3: 185-187.
48. Krapf R, Jaeger P, Hulter HN, 1992 Chronic respiratory alkalosis induces renal PTH-resistance, hyperphosphatemia and hypocalcemia in humans. Kidney Int 42: 727-734.
Address for correspondence:
Peter D. Papapetrou, M.D., Honorary Director, Second Division of Endocrinology, Alexandra Hospital.
51 Panagiotou Street, Papagou, 15669, Attica, Greece, Fax: 210 65 37467, e-mail address: pdpap@otenet.gr
Received 06-11-09, Revised 15-02-10, Accepted 01-03-10